|
12/12/2022 0 Comments Atomic mass definition 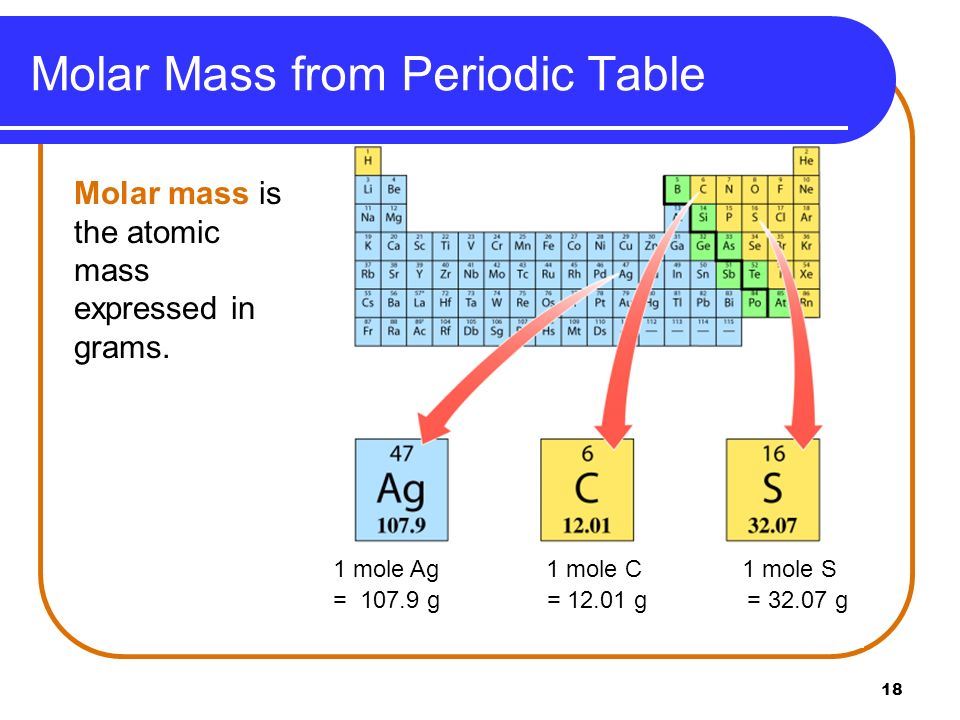 For instance, there is a proposal under consideration to define the kilogram in terms of a specified number of free carbon-12 ( 12C) atoms, at rest and in their ground state, thereby fixing the Avagadro constant, and making an experimental determination unnecessary. It is hoped that in the future the experimental accuracy of Avogadro's constant will improve so much that the unified atomic mass unit may replace the kilogram as the SI base unit. Where the symbol mol represents the mole, which can be defined for any substance as a sample containing the same number of "elementary entities" (atoms or molecules, for example) as there are atoms of 12C in 0.012 kg of carbon-12.įrom this value for N A follows the latest value of the unified atomic mass unit: 1 u ≈ 1.660 538 921(73) × 10 −27 kgįuture refinements in Avogadro's number by future improvements in counting large (on the order of 10 27) numbers of atoms, will give better accuracy of u. 
The latest value of N A is the same numerical value as the Avagadro constant, the number of "elementary entities" in a mole of substance: N A = 6.022 141 29(27) × 10 23 / mol, By the definition of Avogadro's number, the mass of N A carbon-12 atoms, at rest and in their ground state, is 12 gram ( = 12×10 −3 kg). The relationship of the unified atomic mass unit to the macroscopic SI base unit of mass, the kilogram, is given by Avogadro's number N A. By definition it is one twelfth of the mass of an unbound carbon-12 ( 12C) atom, at rest and in its ground state. 
The unified atomic mass unit ( u), or dalton ( Da), is a unit of atomic and molecular mass. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
